Most peptide degradation doesn't happen during research — it happens during storage. Heat, moisture, light, and physical agitation all break peptide bonds, and the damage is invisible until you're already working with a compromised vial. The guidelines below cover both stages: keeping lyophilised powder stable before you open it, and extending the shelf life of a reconstituted solution.
Quick Reference
- Lyophilised, short-term (≤3 months): fridge at 2–8°C, sealed, away from light and the door
- Lyophilised, long-term (3+ months): freezer at −20°C, sealed, in a dark container
- Reconstituted in BAC water: fridge at 2–8°C, protected from light, use within 4–6 weeks
- Never: shake or vortex, leave reconstituted vials at room temperature, freeze a reconstituted solution unless unavoidable
- Always: warm frozen vials to room temperature before opening, use a fresh needle for each draw, discard if appearance changes
Why Peptides Degrade
Understanding the mechanism helps you understand why each storage rule exists:
- Hydrolysis — water cleaves peptide bonds into fragments. Accelerated by heat and pH extremes.
- Oxidation — oxygen attacks residues like methionine, cysteine, and tryptophan, permanently altering the molecule.
- Aggregation — peptides clump into insoluble masses under heat or agitation, reducing bioavailability.
- Deamidation — asparagine and glutamine residues spontaneously convert, changing the charge and activity of the compound.
Lyophilisation (freeze-drying) removes water and dramatically slows all of these pathways. This is why research-grade peptides arrive as powder rather than pre-dissolved solution — the dry state is far more stable for shipping and storage.
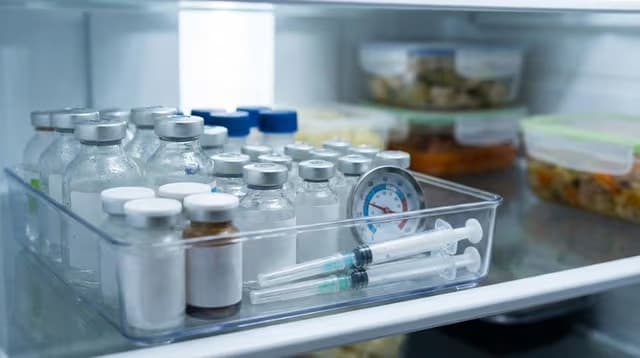
Storing Lyophilised Peptides
Lyophilised peptides are resilient, but not indefinitely so. Heat, moisture, and light still cause damage over time — the goal is to limit all three while keeping the seal intact.
Up to 3 Months
A standard refrigerator at 2–8°C is sufficient for most lyophilised peptides over a 2–3 month window. Store vials in their original packaging or a dedicated container, away from the door — which experiences the most temperature fluctuation with each opening.
3 Months or Longer
For extended storage, a freezer at −20°C is the right approach. Most lyophilised peptides remain stable for 12–24 months or longer at this temperature. Peptides with particularly sensitive residues (cysteine-containing sequences, for example) benefit from −80°C, though this is rarely necessary under standard research timelines.
Rules That Apply at Both Temperatures
- Keep vials sealed. The rubber stopper and aluminium crimp form an airtight barrier. Don't remove it until you're ready to reconstitute.
- Limit freeze-thaw cycles. Each warming-and-cooling cycle introduces condensation risk. If you're storing long-term, freeze and leave frozen — don't repeatedly bring vials to room temperature and return them.
- Block light exposure. UV damages sensitive residues even through glass. Use a dark container, drawer, or opaque bag. Be aware of freezers with internal lights that activate on opening.
- Warm before opening. Take the vial out and let it reach room temperature before reconstituting. This prevents condensation from forming on the powder.
- Store upright. Minimises the surface area of powder in contact with trace gases under the crimp.
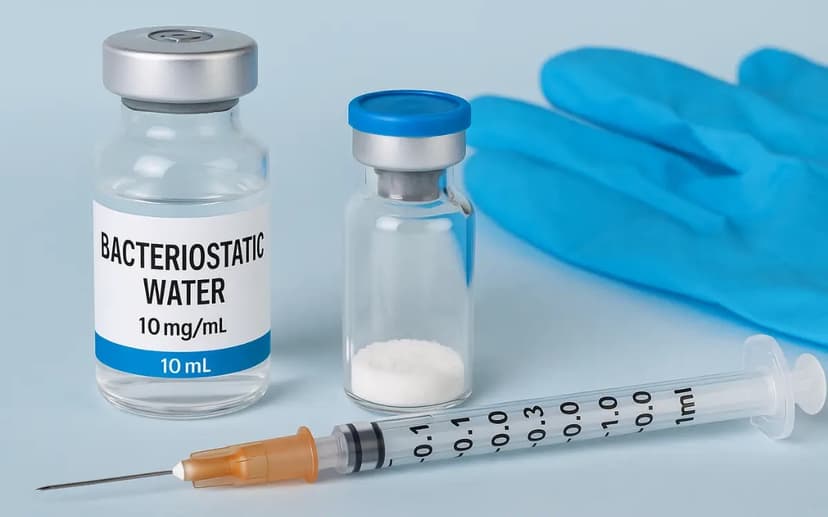
Reconstitution
Reconstitution dissolves lyophilised powder into a liquid carrier. Once this happens, degradation begins — the goal becomes slowing it down, not preventing it.
What to Use
- Bacteriostatic water (BAC water) — the standard for most peptides. Contains 0.9% benzyl alcohol to inhibit microbial growth, enabling multi-dose use over several weeks.
- Sterile water for injection — no preservative. Treat reconstituted vials as single-use or use within 24 hours.
- Dilute acetic acid (0.1%–1%) — required for hydrophobic or poorly soluble peptides such as BPC-157 and TB-500. Use only when the compound specifically calls for it.
- Tap water and distilled water — not sterile. Don't use either.
Technique
Direct the BAC water down the inner glass wall of the vial, not onto the powder. Forceful contact with dry peptide can mechanically disrupt structure. Once the water is in, swirl gently in slow circles — never vortex or shake. Agitation causes aggregation and can denature the compound. Allow a minute or two for complete dissolution; a clear solution is what you're looking for.
Storing Reconstituted Peptides
Reconstitution re-introduces the aqueous environment that lyophilisation removed. All degradation pathways become active again.
Temperature Rules
Refrigerate at 2–8°C immediately after reconstitution. Even a few hours at room temperature accelerates degradation in sensitive sequences. Don't freeze a liquid peptide solution unless you have no alternative — ice crystals physically damage peptide structure and cause aggregation on thawing. If freezing is unavoidable, use −20°C, restrict to a single freeze-thaw, and accept some potency loss.
Shelf Life
- BAC water, refrigerated: stable for 4–6 weeks for most peptides (varies by sequence and concentration)
- Sterile water, refrigerated: single-use or within 24 hours
- Room temperature: hours to a few days at best — not a storage option
Practical implication: only reconstitute what your research protocol will use within the next four weeks. If you have a 10 mg vial but will only use half of it, reconstitute 5 mg and leave the rest as lyophilised powder in the freezer.
Protecting the Vial in Use
- Block light. Wrap in foil or store in a dark container inside the fridge. Certain residues degrade under light even through glass.
- Fresh needle per draw. Reusing needles introduces particulate matter and microbial contamination risk with each puncture.
- Inspect before every use. Cloudiness, particles, or colour change indicates degradation or contamination. Discard — don't use it.
- Limit punctures. Pre-draw multiple aliquots with a separate syringe if your protocol allows; each stopper puncture is a contamination event.
Compound-Specific Notes
- BPC-157 — relatively stable, but acetic acid reconstitution ensures complete dissolution. Refrigerate after reconstitution and use within 4 weeks.
- MT-II (Melanotan II) — stable as lyophilised powder. Keep reconstituted solution cold and protected from light.
- Semax — shorter sequence, well-suited to BAC water. Refrigerate and use within 4–6 weeks.
- Retatrutide — longer 11-residue compound. Standard freezer storage for lyophilised form; refrigerate promptly after reconstitution.
Ambiguous research outcomes often trace back to compromised starting material. A degraded peptide doesn't fail visibly — it just produces unreliable data. Storage discipline is one of the few variables in this process that's entirely within your control.
References
- ICH Q1A(R2) — Stability Testing of New Drug Substances and Products, International Council for Harmonisation (2003)
- Manning et al. — Stability of Protein Pharmaceuticals: An Update, Pharmaceutical Research (2010)
- Wang — Instability, Stabilisation, and Formulation of Liquid Protein Pharmaceuticals, International Journal of Pharmaceutics (1999)
- Cleland et al. — The Development of Stable Protein Formulations: A Close Look at Protein Aggregation, Deamidation and Oxidation, Critical Reviews in Therapeutic Drug Carrier Systems (1993)
- Pikal — Mechanisms of Protein Stabilisation During Freeze-Drying and Storage: The Relative Importance of Thermodynamic Stabilisation and Glassy State Relaxation Dynamics, Drugs and the Pharmaceutical Sciences (2002)
- Chang & Pikal — Mechanisms of Protein Stabilisation in the Solid State, Journal of Pharmaceutical Sciences (2009)
- Carpenter et al. — Overlooking Subvisible Particles in Therapeutic Protein Products: Gaps That May Compromise Product Quality, Journal of Pharmaceutical Sciences (2009)
- Krishnamurthy & Manning — The Stability Factor: Importance in Formulation Development, Current Pharmaceutical Biotechnology (2002)
⚠ All information is for educational and research purposes only. For in-vitro research and laboratory use only. Not for human consumption.