The vial-versus-pen question comes up often in retatrutide research discussions. It is a sensible thing to ask. Pens are the format that most people associate with GLP-1 medicines from the pharmacy, so it is natural to wonder why research-grade retatrutide does not arrive in the same format. The short answer is that pens and vials serve different supply contexts, and the research market is not the same thing as the approved pharmaceutical market.
Stability: Why Lyophilised Vials Are the Research Standard
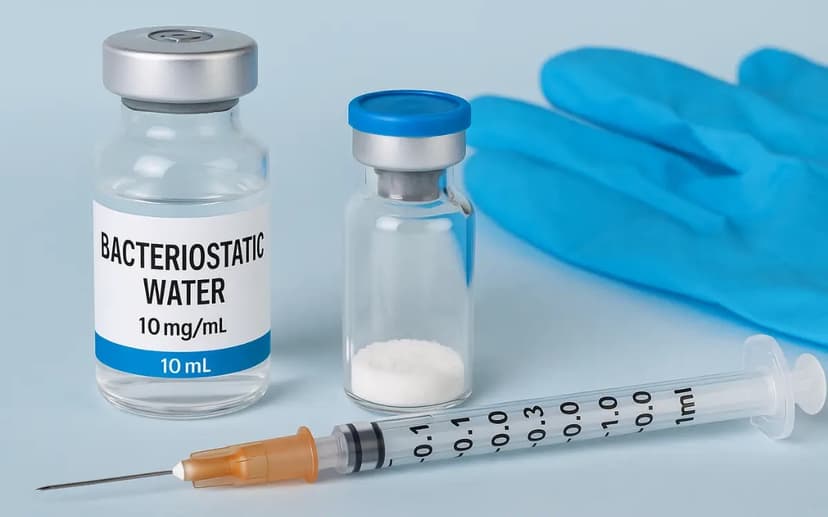
The primary reason research peptides are supplied as dry powder in glass vials comes down to chemical stability. Once a peptide is dissolved in aqueous solution — which is what a pre-filled pen contains — it becomes significantly more susceptible to degradation. Aggregation, oxidation, and hydrolysis all accelerate in the liquid state.
Formulation science literature consistently shows this distinction: lyophilisation is specifically adopted to slow degradation and extend usable shelf life, while liquid formulations require considerably more stabilisation work. [3] [4] [5] For research supply, that makes the lyophilised vial the practical default. It also gives researchers the advantage of:
- Controlled reconstitution volume — flexibility in how concentrated the solution is prepared
- Batch-matched CoA traceability — each vial connects to specific third-party analysis data
- Straightforward cold-chain handling before reconstitution
- No dependence on a fixed consumer injector device
What Makes a Pen Format Work for Approved Medicines
Pre-filled injection pens are engineered for a fundamentally different context. They require a pharmaceutical company to have already developed a validated liquid formulation, a fixed-dose delivery mechanism, a tested device, and a complete regulated prescription supply chain behind them. That combination is what makes a product like Ozempic or Mounjaro pen-deliverable at pharmacy level.
The research-vial model is not trying to replicate that. It is a different supply chain with different tradeoffs. Even within the approved pharmaceutical world, regulators have repeatedly warned about falsified pens circulating outside legitimate channels. The MHRA has issued multiple alerts about unsafe pre-filled weight-loss pens sourced through unregulated routes. [6]
Where Retatrutide Pens Actually Stand Right Now
Lilly's current position is clear: retatrutide is still investigational, remains in Phase 3 clinical trials, and is not yet available for public use. Regulatory approval and commercial launch depend on trial completion and TRIUMPH program outcomes expected through 2026. [1]
That does not mean retatrutide pens have never been seen. In October 2025, the MHRA seized over 2,000 unlicensed retatrutide and tirzepatide pens from an illicit manufacturing site. [2] The accurate picture is:
- Yes, unlicensed retatrutide pens have existed and been seized.
- No, there is no approved public-use retatrutide pen available through legitimate channels.
- No, Aus Peptide Supply does not sell pre-filled retatrutide pens — we supply lyophilised research vials.
Reconstituting a Vial Is Simpler Than It Sounds
A lot of searches for "retatrutide pen Australia" are really about convenience, not the pen specifically. The concern is setup complexity. In practice, the process is not complicated: introduce bacteriostatic water slowly into the vial, allow the powder to dissolve without agitation, store the reconstituted solution under refrigeration, and draw doses using a fine insulin syringe. Our step-by-step reconstitution guide covers the full process clearly if you have not done it before.
Recommended Starting Point
Retatrutide Starter Bundle
If the appeal of a pen is getting everything in one place, the Retatrutide Starter Bundle does the same thing for the vial format. It includes 2× 10mg Retatrutide vials, BAC water, insulin syringes, and alcohol swabs — no need to source each component separately.
The Practical Takeaway
The vial and the pen are not competing quality tiers — they are different tools for different systems. Pens belong to finished pharmaceutical products with validated liquid formulations and device approval. Vials are the research standard because lyophilised storage is more stable, more flexible, and more compatible with batch-specific quality documentation.
If the search was "retatrutide pen Australia": no approved public product exists yet. Research-grade retatrutide supplied as a vial is what is currently available and appropriate for laboratory use.
References
- Eli Lilly — What to know about retatrutide (updated December 2025)
- MHRA — Record seizure of illicit weight loss medicines including unlicensed retatrutide pens (24 October 2025)
- Manning et al. — Stability of Protein Pharmaceuticals: An Update, Pharmaceutical Research (2010)
- Wang — Instability, Stabilisation, and Formulation of Liquid Protein Pharmaceuticals, International Journal of Pharmaceutics (1999)
- Krishnamurthy & Manning — The Stability Factor: Importance in Formulation Development, Current Pharmaceutical Biotechnology (2002)
- MHRA — Warnings on unsafe fake weight-loss pens (26 October 2023)
⚠ All information is for educational and research purposes only. Retatrutide supplied by Aus Peptide Supply is for in-vitro laboratory research only and not for human consumption.